Global In-vitro Diagnostics (IVD) Market Size, Share, Trends and Growth Forecast Report By Product (Instruments, Reagents and Kits and Software), IVD Technology (Clinical Chemistry, Hematology, Immunoassays, Coagulation and Hemostasis, Molecular Diagnostics, Microbiology and Other IVD technologies), Application and Region (North America, Europe, Asia-Pacific, Latin America, Middle East and Africa), Industry Analysis From 2025 to 2033
Global In-vitro Diagnostics (IVD) Market Size
The global in-vitro diagnostics (IVD) market was valued at USD 82.40 billion in 2024. The global in-vitro diagnostics (IVD) market size is estimated to be USD 87.01 billion in 2025. The global IVD market is further forecasted to grow to USD 134.55 billion by 2033 at a compound annual growth rate (CAGR) of 5.6% from 2025 to 2033.
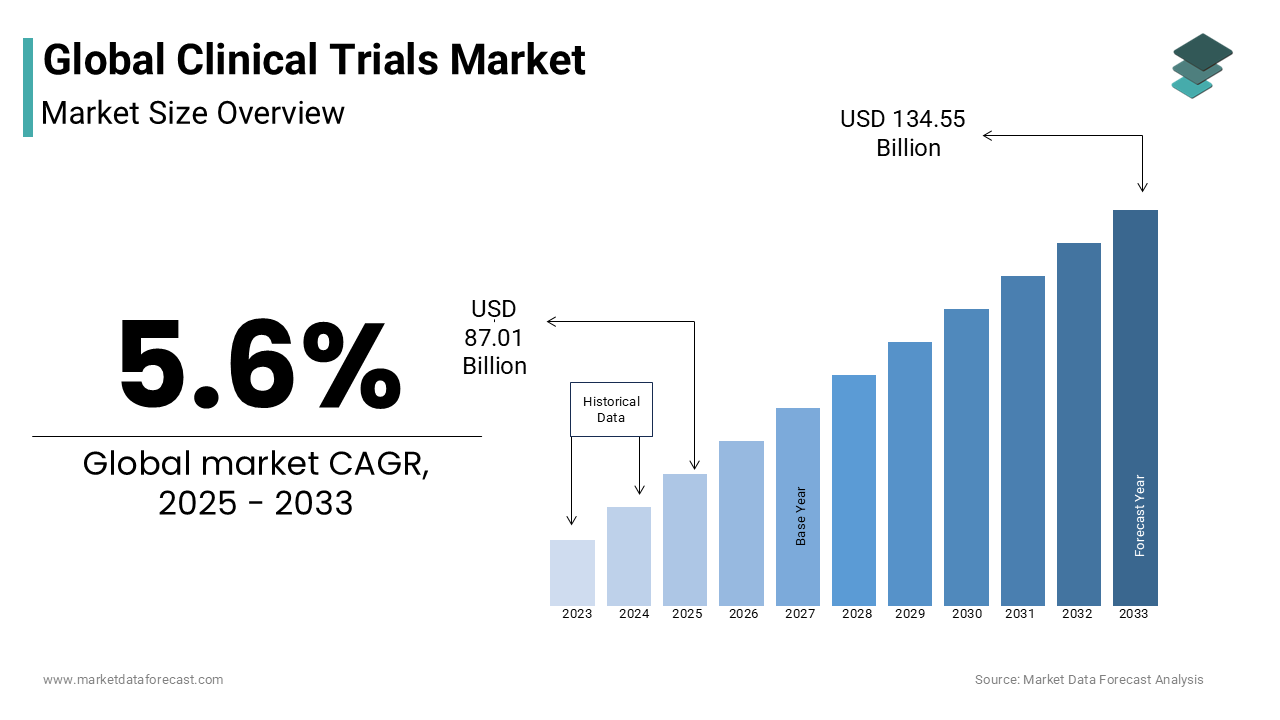
The demand for in-vitro diagnostics (IVD) is on the rise and is expected to accelerate during the forecast period. Factors such as the growing patient population suffering from chronic and infectious diseases, technological advancements, and an increasing aging population are fuelling the need for IVD worldwide. Regions such as North America, Europe and Asia-Pacific have been experiencing substantial demand for IVD products and services and account for the majority of the global IVD market share. The market participants have been adopting strategies such as innovating products with enhanced capabilities, entering new markets through partnerships and acquisitions, and increasing the number of awareness campaigns among healthcare professionals and consumers to increase their market share and strengthen their position in the global market. In addition to these, the key market participants have been exploring strategies such as customization, cost-effectiveness, and focus on personalized healthcare to address the increasing clinical needs and expand access to IVD technologies. Regions such as North America and Europe have been putting increased focus on precision medicine and preventive healthcare, which are resulting in the increased demand for IVD.
MARKET DRIVERS
Rising Prevalence of Chronic and Infectious Diseases
The increasing global burden of chronic and infectious diseases is a major driver of the global IVD market. According to the World Health Organization (WHO), more than 17 million people die annually from cardiovascular diseases, which is resulting in the increasing need for early detection through diagnostics. Similarly, the ongoing threat of infectious diseases like HIV, tuberculosis, and COVID-19 amplifies the demand for diagnostic solutions. IVD tools, such as immunoassays and molecular diagnostics, enable early and accurate detection and improve patient outcomes. As healthcare systems emphasize preventive care, the demand for comprehensive IVD solutions continues to rise to support timely interventions and better management of diseases.
Technological Advancements in Diagnostics
Advances in diagnostic technologies, including next-generation sequencing (NGS), digital PCR, and lab-on-a-chip devices are transforming the IVD market worldwide. These innovations offer high accuracy, reduced turnaround times, and greater accessibility. For instance, NGS is revolutionizing cancer diagnostics by enabling detailed genomic profiling and aiding personalized treatments. According to a 2022 study, 65% of oncologists rely on molecular diagnostics for precise cancer management. Additionally, the integration of artificial intelligence (AI) in diagnostics improves data interpretation and is driving growth in automated and point-of-care testing solutions across various healthcare settings.
MARKET RESTRAINTS
High Costs of Advanced Diagnostic Tools
The high costs associated with advanced IVD technologies, such as next-generation sequencing (NGS) and digital PCR, pose a significant restraint, especially in low- and middle-income countries. For instance, the average cost of an NGS test ranges from $1,000 to $5,000 and is limiting its accessibility to a broader patient population. Additionally, the expenses related to equipment maintenance, reagents, and skilled workforce further increase the financial burden on healthcare providers. These high costs create barriers to adoption in resource-constrained settings, where affordability remains a critical factor in implementing diagnostic solutions.
Regulatory and Compliance Challenges
The stringent and varying regulatory requirements for IVD products across different regions create hurdles for manufacturers. Each country’s distinct approval processes, such as the FDA in the U.S. or the CE marking in Europe, can lead to delays in product launches. For instance, a 2022 survey revealed that 30% of diagnostic companies face regulatory hurdles when expanding to new markets. The need to meet specific regional standards promote operational complexities and costs and slows down innovation and global market penetration for IVD solutions.
MARKET OPPORTUNITIES
Expansion of Point-of-Care Testing (POCT)
The growing demand for point-of-care testing (POCT) presents a significant opportunity in the IVD market. POCT devices that offer rapid results at the site of care are gaining traction in rural and underserved regions. For example, portable glucose meters and COVID-19 rapid antigen tests have revolutionized accessibility. According to a 2023 report, 40% of diagnostic tests in low-resource settings now utilize POCT technologies. The continued development of compact, user-friendly and cost-effective devices provides opportunities to improve healthcare delivery globally, particularly in regions with limited laboratory infrastructure.
Integration of Artificial Intelligence (AI) in Diagnostics
Artificial intelligence (AI) is transforming the IVD landscape by enhancing data analysis and decision-making processes. AI-driven platforms can process vast datasets, identify patterns, and provide precise diagnostic insights. For instance, AI-enabled tools are used in cancer screening to detect early-stage tumors with improved accuracy. As per a 2022 study, AI integration reduces diagnostic errors by 30% and enables faster and more accurate clinical decisions. The increasing adoption of AI-powered diagnostic systems opens new avenues for innovation and fuels the efficiency and outcomes in healthcare settings worldwide.
MARKET CHALLENGES
Data Management and Interoperability Issues
The integration of IVD systems with existing healthcare IT infrastructures presents a significant challenge. Many healthcare facilities struggle with interoperability between diagnostic devices and electronic health records (EHRs), which results in inefficiencies and data silos. As per a 2023 survey, approximately 40% of laboratories face difficulties in standardizing data exchange across platforms. This lack of seamless integration not only delays diagnostic workflows but also hampers the implementation of advanced technologies such as AI.
Skilled Workforce Shortage
A shortage of trained professionals capable of operating advanced diagnostic tools limits the effective utilization of IVD technologies. Techniques such as next-generation sequencing (NGS) and digital PCR require specialized knowledge, which is often lacking in resource-limited regions. According to a 2022 report, 35% of diagnostic laboratories worldwide face workforce shortages and impacting their ability to deliver timely and accurate results. This challenge is particularly pronounced in emerging markets, where training programs and access to education for diagnostic professionals remain inadequate. Bridging this skills gap is critical for expanding the adoption of IVD solutions globally.
REPORT COVERAGE
|
REPORT METRIC |
DETAILS |
|
Market Size Available |
2024 to 2033 |
|
Base Year |
2024 |
|
Forecast Period |
2025 to 2033 |
|
Segments Covered |
By By Product, Technology, Application, and Region |
|
Various Analyses Covered |
Global, Regional & Country Level Analysis; Segment-Level Analysis, Drivers, Restraints, Opportunities, Challenges; PESTLE Analysis; Porter's Forces Analysis; Competitive Landscape; Analyst Overview of Investment Opportunities |
|
Regions Covered |
North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
|
Market Leader Profiled |
Abbott Laboratories, Johnson and Johnson, Siemens Healthcare, Becton Dickinson, Roche Diagnostics, Beckman Coulter Inc., Biomérieux, Ortho Clinical Diagnostics, Inc., Bio-Rad Laboratories Danaher Corporation, Sysmex Corporation, and Thermo Fisher Scientific, Inc. |
SEGMENTAL ANALYSIS
By Product Insights
In the global vitro diagnostics (IVD) market, the reagents & kits segment led the market by capturing 53.8% of the global market in 2023. The dominance of the reagents and kits segment is attributed to the high consumption rates of reagents and kits in various diagnostic procedures, including immunoassays and molecular diagnostics. The continuous need for these consumables in routine testing and disease monitoring and the increasing prevalence of chronic diseases drive the growth of the reagents and kits segment in the global market. Additionally, the rise in self-testing and point-of-care diagnostics has further boosted the utilization of reagents and kits, solidifying the leading position of the segment in the market.
The instruments segment is projected to experience the fastest CAGR of 7.1% over the forecast period. This rapid expansion of the instruments segment is primarily driven by technological advancements in diagnostic instruments, such as automation and integration of artificial intelligence, enhancing accuracy and efficiency in laboratories. The increasing adoption of sophisticated diagnostic equipment in emerging markets, coupled with the need for high-throughput systems in large healthcare facilities are contributing to the growth of the instruments segment in the worldwide market.
By Technology Insights
The clinical chemistry segment held 26.8% of the global in-vitro diagnostics (IVD) market in 2023. The growing chronic disease patient population primarily drives segmental growth. People suffering from chronic diseases such as diabetes, cardiovascular disease, and kidney disease often require regular monitoring of various biomarkers and chemicals in the blood and use clinical chemistry tests. The rising aging population worldwide is another major factor fuelling the segment’s growth rate. People who are aged are much more likely to get diagnosed with chronic diseases and require regular monitoring of health, including the functioning of kidney and liver and electrolyte levels, to which clinical chemistry tests are used.
The molecular diagnostics segment is predicted to hold a substantial share of the global IVD market in 2023 and is expected to register a notable CAGR during the forecast period. The growing patient population suffering from infectious diseases, genetic diseases, and cancer drives the segmental growth. In addition, the growing adoption of personalized medicine, rising efforts from key market participants on the development of advanced sequencing and amplification technologies, and the rising adoption of molecular diagnostics by clinical laboratories and hospitals fuel the segment's growth rate. Furthermore, growing healthcare expenditure and an increasing number of government initiatives to promote early diagnosis and disease prevention contribute to the growth rate of the segment.
The immunoassay segment is expected to witness a CAGR of 9.66% during the forecast period.
By Application Insights
The oncology/cancer segment is expected to have the fastest CAGR of 9.04% in the worldwide in-vitro diagnostics (IVD) market during the forecast period. Factors such as the growing cancer patient population and increasing demand for personalized medicine and targeted therapies majorly drive segmental growth. In addition, growing government funding for cancer research, increasing awareness regarding early cancer detection among people, and increasing number of technological developments in the advanced molecular diagnostic tests for cancer boost the growth rate of the segment.
The infectious diseases segment accounted for 24.7% of the worldwide IVD market share in 2023 and is expected to grow at a notable CAGR during the forecast period owing to the rising incidence of infectious diseases such as multi-drug resistant infections. The growing demand for early diagnosis and treatment and an increasing number of advancements in diagnostic technologies further accelerate the growth rate of the segment.
REGIONAL ANALYSIS
North America was the largest regional segment in the worldwide market and accounted for 37% of the share of the global market in 2023. During the forecast period, the North American IVD market is also expected to register a promising CAGR owing to the increasing prevalence of chronic diseases such as cancer and respiratory diseases. The presence of well-established healthcare infrastructure, high healthcare expenditure and increasing demand for early and accurate diagnosis fuel the growth rate of the IVD market in North America. Furthermore, the growing adoption of personalized medicine by the North American population and the increasing usage of POC diagnostics favor the growth of the North American market. The presence of major international market participants such as Roche Diagnostics, Abbott Laboratories, and Thermo Fisher Scientific in the North American region is a big plus to the North American regional market growth. The U.S. IVD market had the largest share of the North American market in 2023, followed by Canada. During the forecast period, the U.S. is expected to continue to dominate the North American market and Canada is predicted to grow at a healthy CAGR.
Europe is a potential regional market for in-vitro diagnostics worldwide and is expected to grow at a notable CAGR during the forecast period. The growth of the European in-vitro diagnostics market is driven by the presence of sophisticated healthcare infrastructure, the increasing aging population and the rising demand for POC testing devices. The growing number of initiatives from the European governments for disease prevention, rising awareness among people regarding the benefits of early disease detection and favorable reimbursement policies further boost the growth rate of the European market. The rising adoption of healthcare IT and digital health technologies and the favorable regulatory environment for IVD products and services in the European region further support the growth rate of the European market. The German IVD market held the largest share of the European market in 2023 and is expected to showcase a promising CAGR during the forecast period. On the other hand, the UK and France are predicted to control the largest share of the European market during the forecast period.
The APAC market is the most lucrative regional market globally and is predicted to register the highest CAGR during the forecast period. The growth of the APAC in-vitro diagnostics market is attributed to the increasing chronic disease patient population, a growing number of improvements in the healthcare infrastructure in the APAC countries, rising demand for POC diagnostics and the growing number of initiatives from the governments of APAC countries to promote the healthcare access and affordability fuels the IVD market growth in APAC.
Latin America had a considerable share of the worldwide market in 2023 and is expected to grow at a healthy CAGR during the forecast period. The growing adoption of healthcare IT and increasing usage of telemedicine and remote patient monitoring boost the IVD market in Latin America. In Latin America, the IVD market in Brazil was growing consistently and is forecasted to witness a CAGR of 4.98% during the forecast period. Followed by Brazil, the Mexican IVD market is estimated to register a healthy growth rate during the forecast period.
The Middle East and Africa IVD market will have a slight inclination in the growth rate in the coming years. UAE led this regional market in the recent past, and the UAE market was worth USD 430 million in 2023 and is estimated to grow at a CAGR of 5.32% during the forecast period.
KEY PLAYERS IN THE IVD MARKET
Abbott Laboratories, Johnson and Johnson, Siemens Healthcare, Becton Dickinson, Roche Diagnostics, and Beckman Coulter Inc. are a few of the dominating companies in the global IVD market. Other companies that account for a significant share of the IVD market are Biomérieux, Ortho Clinical Diagnostics, Inc., Bio-Rad Laboratories, Inc., Danaher Corporation, Sysmex Corporation, and Thermo Fisher Scientific, Inc.
RECENT HAPPENINGS IN THE GLOBAL IVD MARKET
- In January 2024, ELITech Group made a significant move in the market by announcing the expansion of its product portfolio and launching the Conformite Europeenne – In-vitro diagnostic regulation (CE-IVDR) certified gastrointestinal bacterial PLUS ELITeMGB kit. This strategic step targets the bacterial pathogens associated with gastrointestinal infections, demonstrating the company's commitment to innovation and market growth.
- In January 2024, F. Hoffman-La Roche Ltd announced its acquisition of LumiraDx, a point-of-care diagnostics platform. The primary aim of the acquisition is to support Roche in accessing LumiraDx's novel technology, which enhances the penetration of point-of-care diagnostics products in the market.
- In July 2023, Siemens announced receiving FDA clearance for its Atellica CI analyzer for immunoassay and clinical chemistry in the global markets.
- In October 2023, Sysmex Corporation announced its collaboration with Fujirebio Holdings, Inc., to launch advanced products for immunoassay among patients.
MARKET SEGMENTATION
This research report on the global in-vitro diagnostics (IVD) market has been segmented and sub-segmented based on the product, technology, application, and region.
By Product
- Instruments
- Reagents and Kits
- Software
By Technology
- Clinical Chemistry
- Hematology
- Immunoassays
- Coagulation and Hemostasis
- Molecular Diagnostics
- Microbiology
- Other IVD technologies
By Application
- Diabetes
- Infectious Diseases
- Oncology/Cancer
- Cardiology
- Nephrology
- Autoimmune Diseases
- Drug Testing
- HIV
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- The Middle East and Africa
Frequently Asked Questions
Which segment by product is predicted to lead the IVD market in the coming years?
Based on the product, the reagents and kits segment is expected to lead in the global in-vitro diagnostics market from 2024 to 2032.
Which segment by application is estimated to dominate in the global IVD market in the coming years?
Based on the application, the cancer segment is growing at a rapid pace and is forecasted to showcase domination over the other segments in the global IVD market from 2024 to 2032.
Which region led the global IVD market in 2023?
Geographically, the North American region dominated the in-vitro diagnostics market in 2023.
Related Reports
Access the study in MULTIPLE FORMATS
Purchase options starting from $ 2500
Didn’t find what you’re looking for?
TALK TO OUR ANALYST TEAM
Need something within your budget?
NO WORRIES! WE GOT YOU COVERED!
Call us on: +1 888 702 9696 (U.S Toll Free)
Write to us: [email protected]
